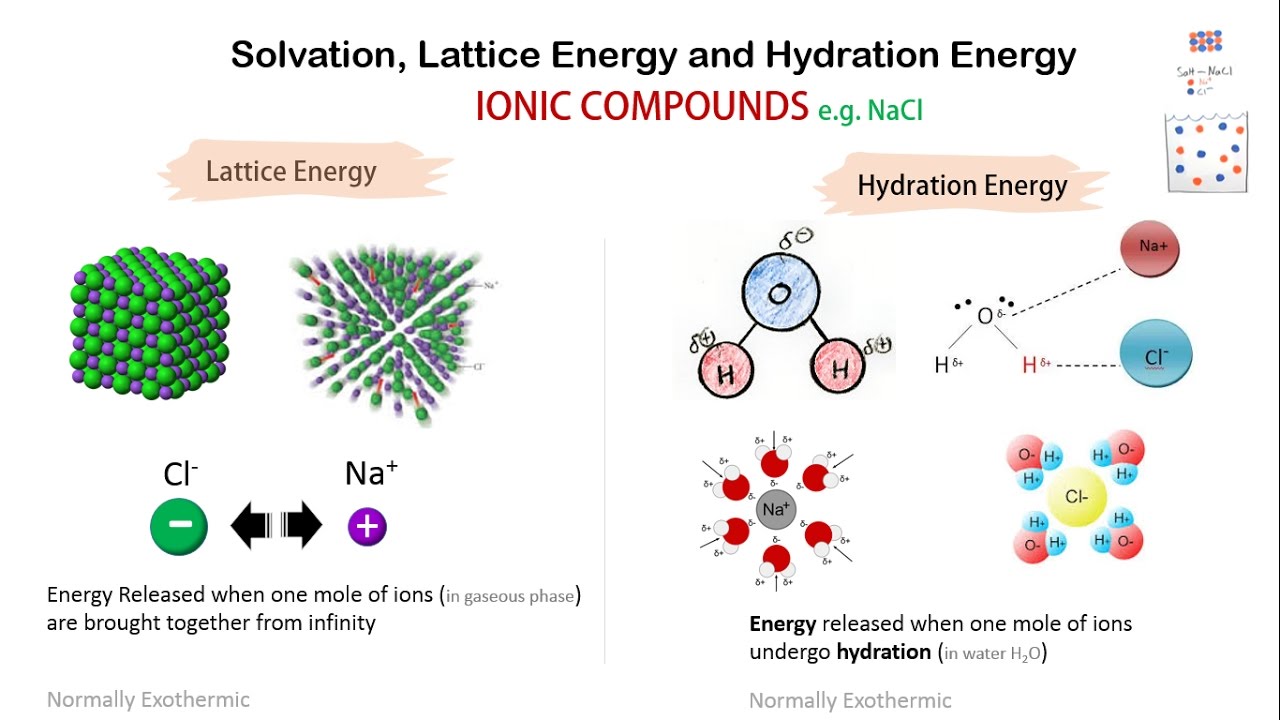
Lattice energy of nacl

This cookie is set by GDPR Cookie Consent plugin. The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. The Born-Lande equation is used to find the lattice energy of a compound, and the formula for the equation is E =. Conversely, for a given alkali metal ion, the fluoride salt always has the highest lattice energy and the iodide salt the lowest. Lattice energies are highest for substances with small, highly charged ions.īecause the ionic radii of the cations decrease in the order K+ > Na+ > Li+ for a given halide ion, the lattice energy decreases smoothly from Li+ to K+. Which one is having highest lattice energy?Ĭonversely, for a given alkali metal ion, the fluoride salt always has the highest lattice energy and the iodide salt the lowest. Third step involved ionisation of sodium in gaseous state to get positively charged sodium ion by losing an electron. The energy of dissociation of bond energy is +122 kJ/mol. Second step involved the dissociation of chlorine molecule to form chlorine atoms in gaseous state.

Sodium fluoride (NaF) shows highest lattice energy among these compounds since Na+ features ions of the same charge, the lattice energy increases as the size of the ions increases.Ĭalculation of lattice energy First step involved the formation of sodium atom in gaseous state from its standard solid sodium. Which shows the highest lattice energy?Īnswer: 1. The smallest ions are at the top of the Periodic Table. They will have the smallest distance between centres and will have the largest lattice energies. The smallest ions will be able to get closest to each other. How do you know which compound has higher lattice energy? As a result, cesium is able to easily donate electrons to the chlorine atom which increases its ionic character. In CsCl, there is high ionic character due to which it has high lattice energy. Which of the following will have the highest lattice energy LiCl mgcl2 NaCl CsCl? This will release the energy equal to lattice energy.

Finally, add the lithium and chloride ion to form a LiCl molecule. This process will be exothermic so, it will be -349 kJ/mol. The chlorine atom accepts an electron and convert into chloride ion by releasing the energy depicted as electron affinity. As implied in the definition, this process will always be exothermic, and thus the value for lattice energy will be negative. The other definition says that lattice energy is the reverse process, meaning it is the energy released when gaseous ions bind to form an ionic solid. What is the lattice enthalpy of NaCl? Why lattice energy is always negative? This results in a greater lattice enthalpy.įor NaCl, the lattice formation enthalpy is -787 kJ mol-1. Because the Li+ ion is smaller than the Na+ ion, the Coulombic attractions between ions in LiCl are stronger than in NaCl. Which has more lattice energy NaCl or LiCl? The negative sign of the energy is indicative of an exothermic reaction. Why is lattice enthalpy of NaCl negative?Īs an example, the lattice energy of sodium chloride, NaCl, is the energy released when gaseous Na+ and Cl– ions come together to form a lattice of alternating ions in the NaCl crystal. ∴ Lattice enthalpy of NaCl = +788.0 kJ mol-1. Lattice enthalpy value from ∆ H0(5) is written with a reversed sign. How do you find the lattice energy of NaCl? 8 What is the formula for lattice energy?.7 Which one is having highest lattice energy?.6 Which shows the highest lattice energy?.5 Which of the following will have the highest lattice energy LiCl mgcl2 NaCl CsCl?.4 Why lattice energy is always negative?.2 Which has more lattice energy NaCl or LiCl?.